2,4-Oxazolidinedione
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name 1,3-Oxazolidine-2,4-dione | |
| Other names 2,4-Oxazolidenedione | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
InChI
| |
| |
| Properties | |
Chemical formula | C3H3NO3 |
| Molar mass | 101.061 g·mol−1 |
| Appearance | white solid |
| Melting point | 89–90 °C (192–194 °F; 362–363 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).  Y verify (what is Y verify (what is  Y Y N ?) N ?) Infobox references | |
Chemical compound
2,4-Oxazolidinedione is an organic compound with the formula HN(CO)2OCH2. It is a white solid. The parent ring is not particularly important, but this core structure is found in a variety anticonvulsant drugs. The parent compound is obtained by treating chloroacetamide with bicarbonate.[2]
-
 Dimethadione
Dimethadione -
 Ethadione
Ethadione -
 Paramethadione
Paramethadione -
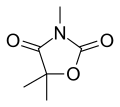 Trimethadione
Trimethadione
See also
- Glycine N-carboxyanhydride, the parent 2,5-oxazolidinedione
References
- ^ Traube, Wilhelm; Ascher, Richard (1913). "Über das Isohydantoin 2-Imino-4-keto-tetrahydro-oxazol und seine Homologen" [About the isohydantoin (?) of 2-imino-4-keto-tetrahydro-oxazole and its homologues]. Berichte der Deutschen Chemischen Gesellschaft (in German). 46 (2): 2077–2084. doi:10.1002/cber.191304602124.
- ^ Cesa, Stefania; Mucciante, Vittoria; Rossi, Leucio (1999). "Tetraethylammonium hydrogen carbonate in organic synthesis: Synthesis of oxazolidine-2,4-diones". Tetrahedron. 55: 193–200. doi:10.1016/S0040-4020(98)01025-4.
- v
- t
- e
modulators
| CA inhibitors |
|
|---|---|
| Others |
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
 | This anticonvulsant-related article is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e
 | This article about a heterocyclic compound is a stub. You can help Wikipedia by expanding it. |
- v
- t
- e















